Clinical Trials on Biofeedback Therapy
New data, new momentum, and new hope for people living with headaches. In this article, we share the results from our latest migraine clinical trial, reflect on what they mean for headache care, and introduce the next study already on the horizon.
This article summarizes the findings from our latest clinical trial. You can read the full press release here.
New migraine trial results offer hope - tension headache study launching next
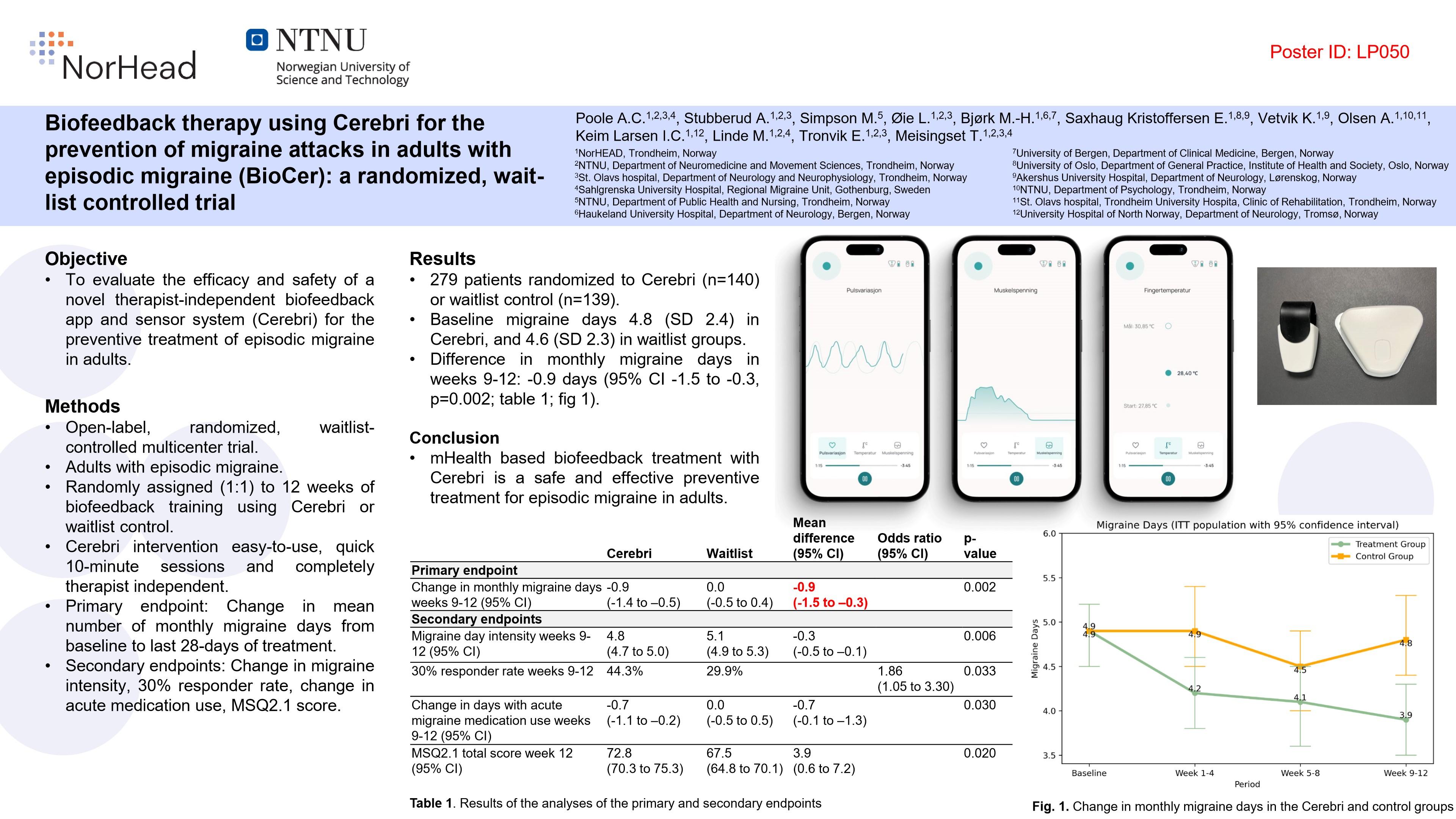
We’re excited to share a major milestone in our journey to transform headache care. Following the successful completion of a randomized controlled trial (RCT) on episodic migraines, our biofeedback technology has shown promising clinical results that are now gaining media attention.
The trial, which ran from January 2023 to September 2024, built on findings from a pilot study conducted in the summer of 2022. In collaboration with our clinical partners, we evaluated our non-invasive, user-friendly biofeedback device on migraine frequency, duration, and overall quality of life.
Early media coverage has highlighted positive outcomes, with participants reporting measurable improvements in their symptoms. While the full research article is currently under review for journal publication, we are proud to see the study already generating discussion among clinicians and patient communities.
You can read one of the media articles covering the results here.
Seeing these results shared publicly is a huge step—not just for us as a company, but for everyone affected by migraines. We set out to create a tool that empowers individuals to better understand and manage their symptoms. This trial validates that mission.
What’s next: Tension-type headache study begins august 2025
Looking ahead, we’re pleased to announce that a new clinical trial focused on tension-type headaches will begin this August. Using the same biofeedback platform, this study aims to explore how our technology can support individuals with one of the most common yet under-treated forms of chronic headache.
“Our next trial will help us broaden the therapeutic reach of biofeedback,” said Jenny Nesje, QA Manager at Nordic Brain Tech. “With the data we’ve gathered so far, we believe we’re just scratching the surface of what this technology can do.”
Recruitment for the new study begins in August 2025. For those interested in participating or following our research, we’ll continue to share updates right here on our site.
Stay tuned as we move forward—expanding the possibilities of biofeedback and building a future where fewer people live under the shadow of chronic headaches.
Bringing this technology home: Introducing Cenli
While Cerebri is being developed as a medical device for clinical use, the same core approach—supporting the nervous system through biofeedback and breathing—is also being made available to the public through Cenli, our upcoming wellness product.
Cenli is a home-based biofeedback tool designed to help people manage everyday stress, improve relaxation, and build focus. It uses similar sensor technology to measure nervous system signals like heart rate variability, finger temperature, and muscle tension, and gives real-time feedback through a simple, app-guided experience.
Cenli is not a medical device and is not intended for the treatment of migraine. However, it reflects our broader mission: making evidence-informed tools for nervous system support more accessible to everyone.
Cenli will launch in fall 2025. Join the waitlist here to get early access and updates.